|
Certain energy changes can be measured experimentally. Combustion means 'burning'. This is one of the energy changes that lends itself to energetics studies. Syllabus reference Syllabus referenceReactivity 2.1.1 - Chemical equations show the ratio of reactants and products in a reaction.
Guidance
Tools and links
Reactivity 1.3.1 - Reactive metals, non-metals and organic compounds undergo combustion reactions when heated in oxygen.
Guidance Tools and links
|
Combustion
Combustion is the term given to burning a substance in air or oxygen. Combustion may be complete, in which case the products are completely oxidised as far as possible by the oxygen, or partial, in which case the products are not completely oxidised. Partial oxidation takes place in a limited air supply.
Complete combustion of methane
CH4 + 2O2 → CO2 + H2O
Compounds containing carbon and hydrogen, i.e. organic compounds, always produce carbon dioxide and water on complete combustion. Incomplete or partial combustion results in the formation of carbon monoxide or even carbon microparticulates.
Incomplete combustion of ethene
C2H4 + 2O2 → 2CO + 2H2O
There isn't only one equation for incomplete combustion, as there are many possible combinations of products.
The standard enthalpy of combustion
The energy released when one mole of a substance is burned in excess oxygen,
or air, under standard conditions. It is given the symbol ΔHc![]() .
.
|
Example: The enthalpy of combustion of ethene may be represented by the equation: C2H4(g) + 2O2(g) →
2CO2(g) + 2H2O(l) ΔH |
REM Combustion always releases energy. It is an exothermic reaction.
Balancing energetics equations
The definition of standard enthalpy change is per mole of reactant, or product, depending on the specific energy being defined).
As energy is an extensive property, it depends on the amount of substance involved and the equations used must reflect only one mole of reactant. This can lead to fractions in the balaced equation. However, it is understood that we are dealing with energetics equations and perfectly acceptable.
|
Example: Show the equation that represents the standard enthalpy change of combustion of hydrogen: Hydrogen has the formula H2, this represents 1 mole of hydrogen. H2(g) + ½O2(g)
→ H2O(l) ΔH Note that the state symbols are given for 25ºC, so that water is a liquid. State symbols must always be included in energetics equations, as there is an energy change requirement when substances move from one state to another. |
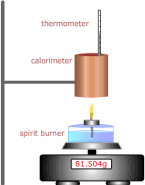
The enthalpy of combustion can be found by burning a substance under controlled conditions and using the energy released to heat a known mass of water.
There are much more accurate methods involving calorimeters (literally 'heat measurers'), which try to take into account the energy lost by radiation and other heat transfers. The data shown in books is from many experiments over the years in which the experimenters have tried to minimise the errors and now agree (within limits) to the values.
This method of enthalpy determination is riddled with errors and inaccuracies, but good enough to demonstrate the procedure.
- 1 Weigh the copper calorimeter (it's really just a copper can!) before. m1, and after filling with a quantity of water m2.
- 2 Weigh the spirit burner before the experiment. m3 - this time in grams
- 3 Record the temperature of the water in the can (calorimeter).
- 4 Light the spirit burner and allow the temperature to rise by at least 20ºC stirring gently to mix thoroughly.
- 5 Weigh the spirit burner after the experiment in grams again. m4.
|
Mass of the water = m2 - m1 kg The energy change in the water = Mass (kg) x specific heat capacity of water x temperature rise (ºC) energy change = mcΔT This gives the energy change for the mass of liquid burned. To calculate the energy change per mole: Mass of liquid burned = m4 - m3 g Moles = mass of liquid burned/ relative molecular mass of the liquid Energy change per mole = energy calculated above/number of moles burned ΔHc = Energy change per mole |
Errors and inaccuracies
The major error in this setup is heat loss by radiation, convection and conduction. The flame only transfers a fraction of its energy to the water. Some energy is radiated away, some energy is used to heat up the can itself, some energy is used to heat up the air, some energy is used to heat up the thermometer etc.
Possible improvements
There are several ways that the experiment can be improved.
- Perform the experiment using an inflammable liquid with a known enthalpy of combustion and work out the percentage efficiency of the apparatus. Use this value to compensate for losses in other experiments.
- Try to find the specific heat capacity of the copper can and use its mass to calculate the energy loss in heating it up.
- Shield the apparatus from draughts.
- Plot a cooling curve of water in the can to find heat loss from the water by radiation.
The bomb calorimeter (not included in the Syllabus requirement) is a means of more accurately measuring enthalpies of combustion. It is recommended to understand the principles involved, to further the appreciation and understanding of the errors inherent in simple laboratory experiments.
Basically, the apparatus consists of a sealed chamber into which a known mass of an inflammable substance is introduced, along with excess oxygen. The heat capacity of the whole apparatus is known, to take into account in the calculations.
The explosive mixture is ignited by a small spark applied electronically using a known amount of electrical energy. The mixture burns rapidly and all of the heat energy released increases the temperature of the apparatus.
The energy released is calculated using:
E = (heat capacity of the calorimeter x ΔT)
The Standard combustion enthalpy is then calculated from:
ΔH![]() = E/moles
= E/moles
Combustion is a process which lends itself to energetics studies and consequently most of the inflammable substances dealt with in chemistry have been determined over the years.
Researchers have investigated methods to improve the results and obtain more accurate data.
As we will see in the section on Hess' law, combustion enthalpy data may be used to calculate enthalpy data that is difficult to obtain directly through experiment, such as enthalpy of formation.
Combustion enthalpy data table

