|
Hydrogen bonding is an intermolecular force of electrostatic attraction between hydrogen atoms attached to electronegative atoms on one molecule and the lone pairs of electronegative atoms on another molecule. A hydrogen bond has a strength about one tenth that of a normal covalent bond. |
|
Hydrogen bonding
Hydrogen bonding is a special case of dipole-dipole attractions.
When hydrogen is bonded to nitrogen, oxygen or fluorine, a very strong dipole is formed, making the hydrogen very strongly (partially) positive. This hydrogen is then attracted to the lone pairs on other similar molecules (nitrogen, oxygen and fluorine all have lone pairs) forming a hydrogen bond, which is stronger than dispersion forces or dipole-dipole interactions, but weaker than normal covalent bonding.
The reason for the strength of the positive charge on the hydrogen atoms, lies in the fact that it has only one bonding pair of electrons. Usually this pair is shared equally between hydrogen and the other bonded atom. However, in the case of hydrogen attached to oxygen, the oxygen pulls the electrons towards it leaving the hydrogen 'naked'.
As the hydrogen atom has no more electron shells, effectively it is the nucleus that is revealed. This is extremely small compared to an atom, so the developing partial charge has a much higher charge density than normal.
The group 15, 16 and 17 hydrides
The effect of hydrogen bonding on intermolecular forces can be demonstrated very well by studying the boiling points of the group 16 hydrides. As expected, the general trend is increased boiling point with increased relative molecular mass (as the dispersion force increases. However, water is completely anomalous to the trend, as it has a much higher boiling point than expected.
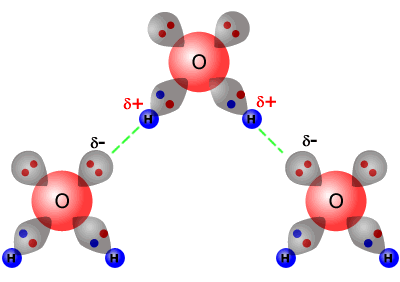 This
anomalous boiling point can be explained by the high degree of hydrogen bonding
between water molecules. Each molecule has two partially positive hydrogen
atoms and it also has two lone pairs of electrons on the oxygen atom.
This
anomalous boiling point can be explained by the high degree of hydrogen bonding
between water molecules. Each molecule has two partially positive hydrogen
atoms and it also has two lone pairs of electrons on the oxygen atom.
This means that the water molecules are able to form two hydrogen bonds per molecule with other water molecules.
The other members of the group 16 hydrides show the effect of increasing relative molecular mass on boiling point. This is to be expected, as the increase in relative molecular mass causes an increase in the strength of the dispersion forces.
Hence there is a gradual but regular increase from H2S to H2Se to H2Te
Comparing molecules
So how do we decide which is the most important force within a molecule? The answer is that we can only compare molecules in which we can 'cancel out' factors. For example, it doesn't make sense to try and compare a very large molecule with only dispersion forces, with a small molecule having hydrogen bonding. Each has a high degree of intermolecular interaction, but they arise from different sources, so can't fairly be compared.
We can, however, compare molecules with similar relative molecular mass, 'cancelling out' the dispersion influence, in which case any difference in boiling point is due to either permanent dipole-dipole interactions, or hydrogen bonding.
|
Example: Compare and explain the boiling points of the molecules CH3OCH3 and C2H5OH. Methoxymethane is a slightly polar molecule, with a partial negative charge on the oxygen. Ethanol is a polar molecule with an O-H group. Both molecules have the same relative mass and so the influence of dispersion forces can be discounted in any comparison. The boiling point of ethanol is much higher than methoxymethane, as ethanol is able to form hydrogen bonds, whereas methoxymethane has rather weaker dipole-dipole interactions. |
Dimerisation
Some molecules have the ability to 'link' to other similar molecules using hydrogen bonding, in such a way that the molecules behave as two in one, so to speak. These double molecules are called dimers.
Ethanoic acid is able to form two hydrogen bonds with another ethanoic acid molecule, making a structure with double the relative molecular mass.
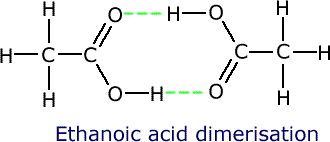
Ethanoic acid has a boiling point of 118ºC, reflecting the high apparent relative molecular mass of 120.
Solubility
The process of dissolution involves the solvent forming bonds with the solute molecules. This bond formation is exothermic, the energy being used to break the interparticular forces holding the structure together. There is also an increase in entropy when a solid dissolves, which contributes to the solubility according to Gibbs Free Energy for the process (section 4.62). If the overall Gibbs Free Energy change is negative, the substance is soluble. (remember: ∆G = ∆H - T∆S)
Substances which hydrogen bond usually dissolve in water. This is because the water molecules themselves can hydrogen bond to the solute. This bond formation makes the process favourable.
Ethanol is an alcohol which is miscible in all proportions with water. This means that it forms hydrogen bonds easily with the water. The solubility of the alcohols decreases as the relative molecular mass increases. As we ascend the homologous series of alcohols the relative mass increases by adding -CH2- units to the hydrocarbon chain.
| alcohol | formula | solubility |
|---|---|---|
| Methanol | CH3OH | miscible |
| Ethanol | CH3CH2OH | miscible |
| Propan-1-ol | CH3CH2CH2OH | miscible |
| Butan-1-ol | CH3CH2CH2CH2OH | soluble |
| Pentan-1-ol | CH3CH2CH2CH2CH2OH | less soluble |
The increasing mass means that dispersion forces become more important and the alcohol molecules begin to form intermolecular bonds with each other more easily than with the water molecules.

