|
Calculations involving weak acids and bases are conditioned by the fact that the acid (or base) does not dissociate 100% into ions. This must be taken into account. |
|
The acid dissociation constant
The acid dissociation constant, Ka, comes from the equilibrium constant for the breakdown of an acid in aqueous solution:
HA + H2O ![]() A- + H3O+
A- + H3O+
Where H3O+ is the hydrogen ion is solution, it may also be written H+(aq). The equilibrium law for this dissociation is:
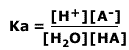 |
As the concentration of water is effectively constant, a new constant, the acid dissociation constant, Ka, is defined as:
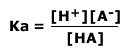 |
The acid dissociation constant, Ka gives a measure of the extent of the dissociation. If Ka is a large value then the acid is strong and dissociates into ions easily. These constants are only useful for weak acids.
|
Acid
|
Ka
|
| Methanoic acid | 1.78 x 10-4 |
| Ethanoic acid | 1.74 x 10-5 |
| Propanoic acid | 1.35 x 10-5 |
| Butanoic acid | 1.51 x 10-5 |
| Hydrocyanic acid | 3.98 x 10-10 |
| Hydrofluoric acid | 5.62 x 10-4 |
| Carbonic acid | 4.26 x 10-7 |
From the list it may be seen that methanoic acid is a stronger acid than ethanoic acid, i.e. it dissociates further releasing more hydrogen ions in solution.
|
Example: Hydrogen ion concentration from Ka.
Ethanoic acid is a weak acid. It only partially dissociates according to the equation:
Therefore the acid dissociation constant: We can assume that as the acid only slightly dissociates then the concentration of the acid at equilibrium is the same (to a close approximation) as the concentration of the original acid (in this case = 0,2 M) Therefore: And as the hydrogen ion concentration equals the ethanoate ion concentration, then: 0.2 x 1.78 x 10-5 = [H+]2 [H+] = √ (3.56 x 10-6) [H+] =1.89 x 10-3 |
pKa values
The pKa values give us a convenient measure of the acid strength. The lower the value the stronger the acid. The pKa value depends on the ease of breakdown of the acid molecule as well as the stability of the ions formed and their relative hydration energies.
The carboxylic acids show a trend over the first three members that is broken with the fourth. The trend from methanoic acid to propanoic acid may be explained by the inductive effect of the alkyl group increasing the electron density in the O-H bond. The reversal of the trend with butanoic acid is possibly due to the lower solubility of the larger hydrocarbon chain of the ion, with corresponding lower hydration enthalpy.
|
Acid
|
pKa
|
| Methanoic acid | 3.75 |
| Ethanoic acid | 4.76 |
| Propanoic acid | 4.87 |
| Butanoic acid | 4.82 |
| Hydrocyanic acid | 9.4 |
| Hydrofluoric acid | 3.25 |
| Carbonic acid | 6.37 |
In this instance you can see that the SMALLER pKa values correspond to the stronger acids. Methanoic acid has a SMALLER pKa value, therefore it is a stronger acid than ethanoic acid.
The base dissociation constant
When bases interact with water they do so by removing a hydrogen ion creating hydroxide ions in solution.
NH3 (aq) + H2O ![]() NH4+(aq) + OH-(aq)
NH4+(aq) + OH-(aq)
Once again, the water concentration is effectively constant and allows us to define a new constant, Kb:
Kb is referred to as the base equilibrium constant and gives a measure of the extent of the equilibrium. Large values for Kb means strong base. However, as for acids, the values are usually very small and the -log(10) form of the information is used to generate convenient sized numbers.
pKb = - log(10) Kb
The lower the pKb value the stronger the base.
Conjugate acids
With bases it may be more convenient to use the conjugate acid of the base to define a new Ka value, this time for the reverse reaction. In the ammonia base equilibrium above, the conjugate acid is the ammonium ion, NH4+, whose dissociation proceeds according to the following equation:
NH4+ + H2O ![]() NH3 + H3O+
NH3 + H3O+
For which the acid dissociation constant is:
This allows us to have a pKa value for a specified base, although it is actually the pKa value of the conjugate acid of the base.
Multiplying Ka and Kb
If the Kb value of a base is multiplied by the Ka value of its conjugate acid we get the following:
Cancelling out the same terms:
Rearrange:
And we already know that [H+][OH-] = Kw = 1.0 x 10-14, therefore:
Take logs and change the sign throughout:
| pKa + pKb = pH + pOH = pKw = 14 |
The following relationships exist:
- pKa + pKb = 14
- pH + pOH = 14
- pH = 14 - pOH
- pKa = 14 - pKb
- pKw = pKa + pKb
- Ka x Kb = Kw
These equations allows us to manipulate acid and base equilibrium constant values very easily.


