|
Optical isomers have an identical arrangement of atoms, but the isomers are mirror images of one another. These mirror images are non-superimposible, just like a pair of gloves. This can only happen if the molecule is asymmetrical. The easiest way for a molecule to be asymmetrical is to have a carbon atom with four different groups or atoms attached. Such an asymmetric carbon atom is called a chiral atom, or centre of chirality. Mirror image optical isomers are called enantiomers. |
|
Physical properties of enantiomers
Optical isomers possess identical physical properties (except in some cases
they form asymmetric mirror image crystals), with one important exception;
a pure optical isomer, or solution of a pure optical isomer rotates the plane
of plane polarised light through an angle. The angle of rotation depends on
the nature of the optical isomer, the solution strength and path length.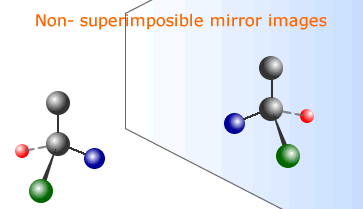
Each optical isomer rotates the plane of polarised light by the same amount, but in the opposite sense, i.e. if one enantiomer rotates polarised light clockwise, then the other enantiomer will rotate plane polarised light through the same angle anticlockwise.
Equal concentrations of two enantiomers do not rotate plane polarised light, as the opposite effects of each optical isomer cancel each other out. This is known as external compensation. Such an externally compensated solution, containing equimolar amounts of two enantiomers, is known as a racemic mixture, or racemate.
Although optical isomerism may seem at first sight to be rather a trivial form of isomerism, it is extremely important in biological systems. The chemical properties of optical isomers are identical EXCEPT when reacting with other optical isomers. This is the basis of the 'key and lock' activation of organic compounds by enzymes.
Biological systems contain many proteins and complex aminoacids, which are optically active. They may be stimulated to react in certain ways by optically active enzymes (biological catalysts).
Chemical properties of enantiomers
The relative positions of all of the component atoms in a pair of optical
isomers are identical. This means that in almost every situation the optical
isomers have identical chemical properties. However, there is one situation
in which two optical isomers can have different reactivity, including one
being totally unreactive while the other is reactive, and this is when they
are reacting with another molecule at a site which itself is asymmetric and
has optical isomers.
The simplest way to appreciate this is to consider the two optical isomers to be a pair of hands, one left hand and one right hand. The reagent which they wish to react with is a left glove. Clearly, the left hand can fit into the left glove, while the right hand cannot.
This principle occurs regularly in biological systems. Many enzymes are chiral and many of the active sites that they bind to are also chiral. The type of optical isomer, + or -, becomes very important. This is sometimes referred to as the lock and key principle. The enzyme is the key and the protein with which it is going to react is the keyhole of the lock. If the key is incorrect it cannot open the lock.
Measuring optical rotation
The 'optical rotation' means the angle through which plane polarised light is turned by the optical isomer under investigation. It is measured using a polarimeter.
An optical isomer may rotate plane polarised light in a clockwise manner (+) or in an anticlockwise manner (-). The isomers are said to be dextrorotatory (rotate to the right) and Laevorotatory (cause rotation to the left). There is no way of knowing how a specific optical isomer will affect plane polarised light just by looking at the structure.
|
|
|
The specific rotation of a chemical compound is defined as the observed angle of optical rotation a when plane-polarised light is passed through a sample with a path length of 10 cm at a sample concentration of 1 g/100cm3.
Classifying enantiomers
There are several systems for classifying the two enantiomeric forms arising from an asymmetric centre. They either depend on the effect that the enantiomer has on plane polarised light, the configuration with respect to a reference molecule or the configuration with respect to a system of priorities Cahn-Ingold-Prelog
The d/l system
An enantiomer can be named by the direction in which it rotates the plane of polarized light.
If it rotates the light clockwise (as seen by a viewer towards whom the light is traveling), that enantiomer is labeled (+). Its mirror-image is labeled (-).
The (+) and (-) isomers have also been termed d- and l-, respectively (for dextrorotatory and levorotatory). Naming with d- and l- is easy to confuse with D- and L- labeling and is therefore strongly discouraged by IUPAC.
The D/L system
An optical isomer can be named by the spatial configuration of its atoms. The D/L system (named after Latin dexter and laevus, right and left), not to be confused with the d- and l-system, does this by relating the molecule to glyceraldehyde.
To determine the D/L isomeric form of an amino acid the "CORN" rule is used. The groups:
COOH, R, NH2 and H (where R is the side-chain)
are arranged around the chiral center carbon atom. With the hydrogen atom away from the viewer, if the arrangement of the CO, R & N groups around the carbon atom is counter-clockwise, then it is the L form.
If the arrangement is clockwise, it is the D form. The L form is the usual one found in natural proteins. For most amino acids, the L form corresponds to an S absolute stereochemistry, but is R instead for certain side-chains.
The R/S system
The R/S system is the most important nomenclature system for denoting enantiomers, which does not involve a reference molecule such as glyceraldehyde.
It labels each chiral center R or S according to a system by which its substituents are each assigned a priority, according to the Cahn–Ingold–Prelog priority rules (CIP), based on atomic number.
If the center is oriented so that the lowest-priority of the four is pointed away from a viewer, the viewer will then see two possibilities:
If the priority of the remaining three substituents decreases in clockwise direction, it is labeled R (for Rectus, Latin for straight), if it decreases in counterclockwise direction, it is S (for Sinister, Latin for left).