|
The hydrocarbons are a massively important group of organic compounds as they constitute the majority of crude oil and natural gas. Hence they are the primary feedstock for the petrochemicals industry. Syllabus reference Syllabus referenceReactivity 1.3.3 - Fossil fuels include coal, crude oil and natural gas, which have different advantages and disadvantages.
Guidance
Tools and links
Reactivity 1.3.4 - Biofuels are produced from the biological fixation of carbon over a short period of time through photosynthesis.
Guidance
Tools and links |
Hydrocarbons
Hydrocarbons are compounds containing only hydrogen and carbon.
There is a huge diversity in the number of compounds that can be formed from only carbon and hydrogen atoms. They may consist of straight chains, branched chains or ring structures; they may contain only single bonds, double bonds, triple bonds, or aromatic systems in which electrons are delocalised over several carbon atoms.
To make sense of this diversity, hydrocarbons can be categorised into three main groups:
- Aliphatic hydrocarbons - straight and branched molecules
- Alicyclic hydrocarbons - ring structures
- Aromatic hydrocarbons - benzene and other delocalised ring structures
These names do not need to be learned, they are for reference only.
Sources of hydrocarbons
Hydrocarbons are mainly derived from fractional distillation and processing of crude oil, other fossil fuel or biotechnological sources. They are used as an energy source and a petrochemical feedstock (raw materials needed to produce other important chemicals and other materials such as plastics and polymers)
Fossil fuels
Literally, combustable materials derived from the breakdown products of organic matter.
Primarily:
- Natural gas
- Crude oil
- Coal
Fossil fuels, including coal, oil, and natural gas, are non-renewable energy sources formed from the decayed remains of plants and animals over millions of years. The type of fossil fuel formed depends on the original material, pressure, heat, and time involved in its formation. Coal, formed primarily from plant material, is mostly carbon with varying amounts of other elements like hydrogen, sulfur, oxygen, and nitrogen. Oil and natural gas, derived from marine organisms, are composed of hydrocarbons with different chain lengths and structures, contributing to their varied physical and chemical properties.
The extraction and use of fossil fuels pose significant environmental challenges. Burning these fuels releases carbon dioxide (CO2), sulfur dioxide (SO2), nitrogen oxides (NOx), and particulate matter into the atmosphere. These emissions contribute to air pollution, acid rain, and health issues. Furthermore, the extraction process itself can lead to habitat destruction, water pollution, and oil spills, causing long-term ecological damage.
One of the most critical issues associated with the use of fossil fuels is their contribution to the greenhouse effect. The greenhouse effect is a natural process where certain gases in Earth's atmosphere trap heat, keeping the planet warm enough to support life. However, human activities, particularly the burning of fossil fuels, have increased the concentration of these greenhouse gases, notably CO2, methane (CH4), and nitrous oxide (N2O), leading to enhanced greenhouse effect. This results in global warming and climate change, with impacts such as rising sea levels, changing weather patterns, and increased frequency of extreme weather events.
Hydrocarbons as an energy source
All hydrocarbons are flammable and produce carbon dioxide and water when burned in excess air or oxygen. Incomplete combustion may produce carbon monoxide and carbon microparticulates (small carbon containing particles). These products of incomplete combustion are a pollution hazard.
All complete combustion reactions can be represented by a general equation:
![]()
This allows investigators to find the formula of any organic hydrocarbon by measuring the carbon dioxide and water it produces on complete combustion.
Biofuels
Biofuels are renewable energy sources derived from biological materials, such as plants or animal waste. They are considered alternatives to fossil fuels and are categorized mainly into two types: bioethanol and biodiesel. Bioethanol, a type of alcohol, is produced primarily through the fermentation of carbohydrates in sugarcane, corn, or other crops rich in sugars or starches. Biodiesel is made through a chemical process called transesterification, where oils and fats (often from soybean, palm, or used cooking oil) are converted into fatty acid methyl esters (FAMEs).
The production of biofuels varies depending on the type and source of biomass. For bioethanol, the process typically involves milling, fermentation, and distillation of feedstock crops. In biodiesel production, the process includes extracting oil from feedstocks and then subjecting it to transesterification, where triglycerides react with an alcohol (usually methanol) in the presence of a catalyst to form biodiesel and glycerol.
Example of a trans-esterification reaction
The transesterification reaction for producing biodiesel can be represented as:
Triglyceride + 3(Methanol) → 3(Fatty Acid Methyl Ester (Biodiesel)) + Glycerol
Chemical equation:
R-COO-CH2-CH(OOCR')-CH2-OOCR'' + 3CH3OH → 3R-COO-CH3 + CH2OH-CHOH-CH2OH
Where R, R', and R'' represent the fatty acid chains, which can vary in length and saturation.
This reaction is typically catalyzed by a strong base or acid, and the glycerol formed as a byproduct has various industrial applications.
Despite being touted as environmentally friendly alternatives to fossil fuels, biofuels have sparked several controversies. One major issue is the "food versus fuel" debate, where the use of food crops for biofuel production is argued to contribute to rising food prices and shortages in some regions. Additionally, the cultivation of crops for biofuels has raised concerns about deforestation, loss of biodiversity, and increased greenhouse gas emissions due to land use changes. Another point of contention is the overall carbon footprint of biofuels, considering the emissions from cultivation, harvesting, processing, and transportation of feedstocks, which may offset some of the environmental benefits.
Aliphatic hydrocarbons
Aliphatic hydrocarbons do not contain delocalised electron systems (benzene rings, primarily). They can be sub-divided into straight chains, branched chains.
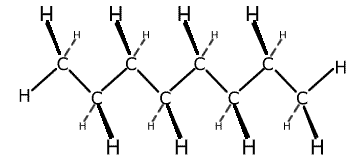
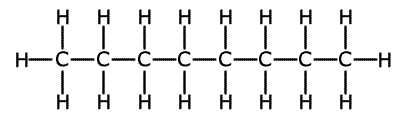
Straight chain hydrocarbons are actually a zig-zag shape, as the bond angle formed between carbon-carbon-carbon atoms is actually 109.5º. However, it is more convenient to represent the structure as though it were straight with right angles. The term 'straight chain' is used to differentiate the structures from branched chain molecules.
This is shown below using the straight chain molecule octane, C8H18.
|
Octane representations
|
|
 |
 |
|
3-dimensional representation
|
2-dimensional representation
|
Although the 2-dimensional (flat) structure is not strictly speaking correct, it contains all of the information that is needed.
Branched chains have carbon containing chains branching off the main chain, just like branches of the main trunk of a tree. Once again, in reality the carbon atoms are all arranged in a zig-zag fashion.
|
2-methyloctane showing the branched
structure
|
|
 |
 |
|
3-dimensional representation
|
2-dimensional representation
|
Alicyclic hydrocarbons
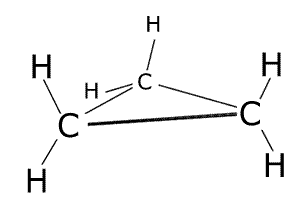
These are molecules that are arranged into ring structures. The prefix cyclo- is used to indicate the ring. The smallest of the hydrocarbon ring structures is cyclopropane, C3H6.
Small cyclic molecules have different chemical properties from their straight chain counterparts. Due to the higher energy of a strained ring structure they are more reactive. The impossibility of rotation about carbon-carbon bonds means that they display geometric isomerism when two or more other atoms or groups are bonded to the ring structure.
|
cyclopropane - C3H6
|
|
 |
 |
|
3-dimensional representation
|
2-dimensional representation
|
Ring structures can get very complicated, with one molecule having several different rings, for example, cubane C8H8.
Unsaturated hydrocarbons
The terms saturation and unsaturation refer to the type of bonding between the carbon atoms in a structure. A molecule that contains only carbon-carbon single bonds is said to be saturated. Double bonds and triple bonds are said to be unsaturated.
|
oct-3-ene
- C8H16
|
|
 |
 |
|
3-dimensional representation
|
2-dimensional representation
|
When a molecule contains many double (or triple) bonds it is said to be polyunsaturated. In long chain hydrocarbons unsaturation gives rise to 'kinks' in the structure and prevents the long chains aligning easily. This lowers the melting point.
The degree of unsaturation can be determined using reagents that react quantitatively with the double bonds.
Aromatic hydrocarbons
The actual definition of aromatic not necessary at this level, but they can be considered to be compounds that contain benzene rings. These have their own very characteristic properties.

