Standard level
In a nucleophilic substitution reaction, a nucleophile donates an electron pair to form a new bond, as another bond breaks producing a leaving group
Syllabus ref: R3.4.2Reactivity 3.4.2 - In a nucleophilic substitution reaction, a nucleophile donates an electron pair to form a new bond, as another bond breaks producing a leaving group.
- Deduce equations with descriptions and explanations of the movement of electron pairs in nucleophilic substitution reactions.

Guidance
- Further details of the mechanisms are not required at SL.
Tools and links
Substitution reactions
Substitution means replacement of one atom or group by another.
Halogenoalkanes are attacked by nucleophilic reagents (reagents seeking a positive charge) and undergo substitution of the halide ion by the nucleophile.
The general reaction scheme is as follows:
R-X + Nu- → R-Nu + X-
Where R is an alkyl chain, X is the halide ion and Nu the nucleophile.
Reaction with hydroxide ions
Halogenoalkanes undergo nucleophilic substitution on warming with dilute alkali, making alcohols:
chloroethane + sodium hydroxide → ethanol + sodium chloride
CH3CH2Cl + NaOH → CH3CH2OH + NaCl
The hydroxide ion is the nucleophile and the chloride ion is said to be the 'leaving group'.
Nucleophilic substitution with cyanide ions, CN-
The cyanide ion, CN- is a good nucleophile and reacts with haloalkanes producing nitriles.
potassium cyanide + bromoethane → ethanonitrile + potassium bromide
KCN + CH3CH2Br → CH3CH2CN + KBr
This is a useful reaction for increasing the chain length by one carbon atom. Nitriles, themselves can be reduced to amines by hydrogen/nickel catalyst at 250ºC.
Mechanisms of nucleophilic substitution
Nucleophilic substitution with cyanide ions
Step 1: The cyanide ion attacks at the partially positive carbon of the dipole, making a high energy transition state.
Step 2: The bromine atom leaves with its bonding electrons as a bromide ion
Step 3. The final product is a nitrile.
Nucleophilic substitution of ammonia, NH3
Ammonia reacts with haloalkanes producing amines. The mechanism once again depends on whether the haloalkane is 1º, 2º or 3.
ammonia + bromoethane → ethylamine + hydrogen bromide
NH3 + CH3CH2Br → CH3CH2NH2 + HBr
Step 1: The nitrogen atom of ammonia has a lone pair that can attack the partially positive carbon, attached to the halogen atom
Step 2: The bromine atom leaves with its bonding electrons as a bromide ion
Step 3. The final product is an amine
Primary, secondary and tertiary haloalkanes
Nucleophilic substitution proceeds via different mechanisms, depending on whether the haloalkane is primary, secondary or tertiary. There are two distinct mechanisms, one for primary and one for tertiary. The mechanism followed by secondary haloalkanes is thought to be a mixture of the other two.
Primary halogenoalkanes
The nucleophile attacks the partially positive carbon that is attached to the halogen atom. This goes through a high energy transition state in which the carbon atom is associated with the incoming nucleophile as well as the outgoing halide ion.
Bimolecular Nucleophilic Substitution
Step 1: The hydroxide ion is attracted towards the partially positive carbon atom.
Step 2: A high energy transition state develops.
Step 3. The chloride ion (leaving group) breaks off leaving the hydroxide ion bonded to the alkyl group.
The mechanism for nucleophilic substitution involves two particles, the haloalkane and a nucleophile in the initial stage of reaction. For this reason it is said to be 'bimolecular'. SN2 stands for substitution - nucleophilic - bimolecular.
Tertiary halogenoalkanes
Tertiary haloalkanes react via a different mechanism. The tertiary carbonium ion formed by loss of a halide ion from the halogenoalkane is sufficiently stable to exist independently. The tertiary halalkane is in equilibrium with this carbonium ion:
2-bromomethylpropane ⇋ tertiary carbonium ion + bromide ion
(CH3)3C-Br ⇋ (CH3)3C+ + Br-
The nucleophile can attack the tertiary carbonium ion as soon as it is formed. The rate determining step is the dissociation of the tertiary haloalkane, which only involves one species. For this reason it is said to be unimolecular. sN1
Unimolecular Nucleophilic Substitution
Step 1: The bromine atom first breaks off as an ion, leaving a tertiary carbonium ion
Step 2: The tertiary carbonium ion is attacked by the hydroxide nuclophile.
Step 3. The final product is a tertiary alcohol.
Summary of nucleophilic substitution in haloalkanes
Factors affecting reaction rate
The rate of reaction is dependent on two factors:
- Whether the haloalkane is primary, secondary of tertiary
- The actual halogen atoms attached to the alkyl chain
Mechanism
Tertiary haloakanes react via an sN1 mechanism that has a much lower activation energy than the sN2 mechanism with the high energy transition state. Hence tertiary haloalkanes react faster then secondary, which in turn react faster than primary.
The halide ion leaving group
The bond fromed between carbon and iodine is much weaker than the bond formed between bromine and carbon, which in turn is weaker than the bond formed between chlorine and carbon. It is therefore easier to break off an iodide ion than a bromide or chloride ion. The rate of reaction is in the order:
iodoalkanes >> bromoalkanes >> chloroalkanes
Note: Students are no longer expected to know the effects of solvent on reaction rate
Stereospecificity
Mechanisms that proceed via an sN2 mechanism cause inversion at the carbon atom that is attacked by the nucleophile.
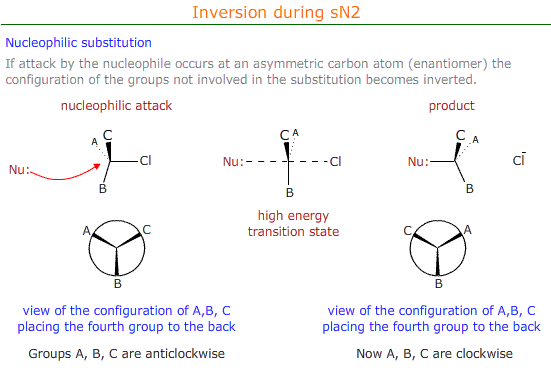
This has consequences for the optical properties and the designation of the configuration (R or S) of the stereoisomer.
If the enantiomer has an "R" conformation before nucleophilic substitution, as determined by CIP priority rules, then the product of the substitution has an "S" configuration