|
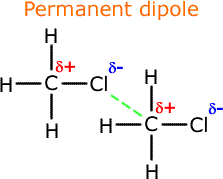
As suggested by the name, these are forces due to permanent dipoles within the molecules. |
|
Polar molecules
|
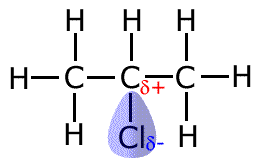
Permanent dipole-dipole interactions occur between molecules that have permanent dipoles. As we have seen in the polarity section, a permanent dipole is created in a molecule when there is an electronegative atom, such as a oxygen, nitrogen or a halogen. The permanent dipole consists of regions of partial positive charge and regions of partial negative charge within the same molecule. By convention we use the small greek letter delta, δ, to represent a partial charge. Hence, δ+ means a partial positive charge and δ- means a partial negative charge. |
 |
Permanent dipole - dipole interactions
In a body of polar molecules the dipoles can attract their oppisite charges on neighbouring molecules. The partial positive charges are attracted to the partial negative charges on other molecules.
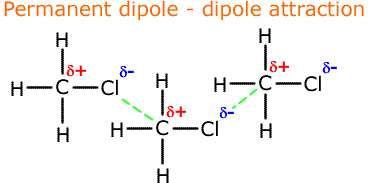 |
The consequence is that the molecules require larger forces to pull them apart. This means that the boiling point and the enthalpy of vaporisation is higher.
Comparison with dispersion forces
You should remember at this point that ALL molecules have dispersion forces. So any comparison must take this into account.
The best way to compare then is to use molecules that have similar relative mass. The vander Waals' forces must be similar in the two types of compound as they are a function of the relative mass. Hence any difference can be thought of as due to the permanent dipole-dipole forces.
Alkanes are examples of compounds that have only dispersion forces.
| Alkanes | Mr | b.p. /ºC | Alkanals | Mr | b.p. /ºC |
|---|---|---|---|---|---|
| ethane | 30 | -89 | methanal | 30 | -21 |
| propane | 42 | -42 | ethanal | 42 | 21 |
| butane | 54 | -0.5 | propanal | 54 | 49 |
We can see from the table that whereas the relative mass is the same between alkanes and alkanals (aldehydes), the boiling point of the alkanals is much higher. This can only be due to some other form of intermolecular force, other than dispersion force. It is of course due to dipole - dipole forces acting between the relatively negative oxygen on the carbonyl group of the alkanal and the relatively positive carbon of the carbonyl group.

