|
As has been shown by Boyle's law and Charles' law, the behaviour of gases conforms (to a close approximation) to some simple relationships between the gas volume, pressure and temperature for a given amount of gas. Combining Boyle's law and Charles' law gives the equation of state. |
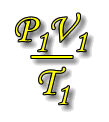 |
The equation of state
The equations of Boyle's law (PV = constant) and Charles' law (V/T = constant) may be combined to give the expression: PV/T = constant. This is called the equation of state. This new combined gas equation can be used to find the new volume, pressure and temperature of a gas if the conditions are changed.
|
The equation of state:
|



